| Identification |
| Name: | N,N'-Dicyclohexylcarbodiimide |
| Synonyms: | DCC; DICYCLOHEXYL CARBODIMIDE; Dicyclohexylcarbodiimide; N,N'-Dicyclohexyl carbodiimide; N,N'-Dicyclohexylcarbodimide |
| CAS: | 538-75-0 |
| EINECS: | 208-704-1 |
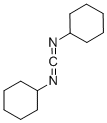
| Molecular Formula: | C13H22N2 |
| Molecular Weight: | 206.33 |
| InChI: | InChI=1/C13H22N2/c1-3-7-12(8-4-1)14-11-15-13-9-5-2-6-10-13/h12-13H,1-10H2 |
| Molecular Structure: |
 |
| Properties |
| Transport: | 20kgs |
| Density: | 1.325 |
| Stability: | Stable, but moisture sensitive. Combustible. Incompatible with strong oxidizing agents. Avoid exposure to air or moisture. |
| Refractive index: | n20/D 1.48 |
| Water Solubility: | Reaction |
| Solubility: | reaction with water |
| Appearance: | colorless solid |
| Specification: |
Dicyclohexylcarbodiimide (CAS NO.538-75-0) is also named as 1,3-Dicyclohexylcarbodiimide ; Bis(cyclohexyl)carbodiimide ; Carbodicyclohexylimide ; DCC ; DCCD ; DCCI ; N,N'-Dicyclohexylcarbodiimide ; N,N'-Methanetetraylbiscyclohexaamine ; NSC 30022 . Dicyclohexylcarbodiimide (CAS NO.538-75-0) is colorless solid with a heavy sweet odor. Dicyclohexylcarbodiimide is an imide. Amides/imides react with azo and diazo compounds to generate toxic gases. Flammable gases are formed by the reaction of organic amides/imides with strong reducing agents. Amides are very weak bases (weaker than water). Imides are less basic yet and in fact react with strong bases to form salts. That is, they can react as acids. Mixing amides with dehydrating agents such as P2O5 or SOCl2 generates the corresponding nitrile. The combustion of these compounds generates mixed oxides of nitrogen (NOx). Dicyclohexylcarbodiimide is incompatible with acids and oxidizing agents. It is probably combustible.
|
| Packinggroup: | II |
| HS Code: | 29252000 |
| Storage Temperature: | Store at RT. |
| Sensitive: | Moisture Sensitive |
| Usage: | This product is mainly used in amikacin, glutathione dehydrants, as well as in synthesis of acid anhydride, aldehyde, ketone, isocyanate; when it is used as dehydrating condensing agent, it reacts to dicyclohexylurea through short-time reaction under normal temperature.
This product can also be used in synthesis of peptide and nucleic acid. It is easy to use this product to react with compound of free carboxy and amino-group into peptide.
This product is widely used in medical, health, make-up and biological products, and other synthetic fields. |
| Safety Data |
| Hazard Symbols |
 Xn:Harmful
Xn:Harmful
|
| |
 |